Methanol
Methanol is a basic, one-carbon molecule that enables a huge number of chemical processes. Made mostly from natural gas, it is at the trailhead of hundreds of value-chain pathways that lead through the realms of petrochemicals, alkalis, thiols and life-sciences chemicals, fundamental to modern-day consumer goods that society demands. Accordingly, methanol is a strategic chemical for a vast array of petrochemical industry participants.
The major uses for methanol are depicted in the table below, demonstrating its versatility. This is only a partial listing (prioritising the major groupings), with many smaller derivatives also comprising the balance.
Major Methanol Derivatives and Uses
Acetic acid
- Vinyl acetate
- Acetic anhydride
- Terephthalic acid
Formaldehyde
- Urea formaldehyde resins
- Melamine resins
- Phenol formaldehyde resins
- Polyoxymethylene (POM or polyacetal)
- Polyols
- Butanediol
- Polyesters (polybutyl terepthalate)
- MDI
- Isoprene
- Rubber (polyisoprene)
- Paraformaldehyde
- Hexamine
Methanol to olefins (MTO)
- Ethylene and derivatives
- Propylene and derivatives
- C4/butadiene
- C5+
- Water
Methyl tert-butyl ether (MTBE)
- Gasoline additives
- Isobutylene
Methyl methacrylate
- Polymethylmethacrylate (PMMA)
- Methacrylate/Acrylate Co Polymers
- Methyl Chloride (Chloromethane)
Developmental Methanol Uses
- Direct uses (gasoline blending)
- Fuel cells
Methylamines
- Monomethylamine
- Caffeine (stimulant, diuretic)
- Sevin/carbaryl (insecticide)
- Various other insecticides
- Various other herbicides
- Various other pesticides
- Water gel explosives
- Photographic developers
- Analgesics (Demerol)
- Antispasmodics
- Dimethylamine
- Fungicides
- Dimethyl formamide (solvent)
- Rubber accelerators, processing agents
- Propellants
- Antihistaminic (Benadryl)
- Catalysts (Urethane)
- Surfactants
- Water treatment
- Detergents
- Germicides
- Herbicides
- Epoxy resin accelerators
- Trimethylamine
- Acid Scavenger (nylon, benzyl esters)
- Choline chloride
- Ion exchange resins (with crosslinked polystyrene)
- Gelling Inhibitor (polyester)
Other Methanol Uses
- Dimethyl terephthalate
- Polyesters
- Reducing agent
- Purified terephthalic acid (PTA)
- Methyl mercaptan (methanethiol)
- Chlorine dioxide
- DL-methionine (amino acid)
Methanol has also been used as an "alternative fuel." In Europe, methanol is used in the production of biodiesel, which can replace refinery-based diesel for use in transportation. In China, methanol is used directly as a blending component of gasoline, driven by the need to extend the octane pool in that country and due to economic feasibility as high crude oil and gasoline prices have prompted the use of cheaper methanol. Methanol has also been considered for direct combustion in combined cycle power generation facilities.
There is also a significant commercialisation effort underway in two developmental uses for methanol: fuel cells and methanol-to-olefins (MTO or gas-to-olefins—GTO). Fuel cells can utilise the hydrogen molecules of methanol (as well as other fuels) to create electricity (and water). MTO/GTO utilise methanol as an intermediary step in the production of olefins and their derivatives (ethylene, propylene, polyethylene, polypropylene). All of these "alternative fuel" uses for methanol face significant hurdles in their commercialisation but high potential demand.
Markets
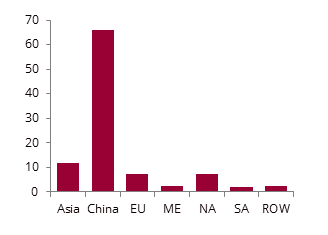
Methanol demand by major region in 2024, %
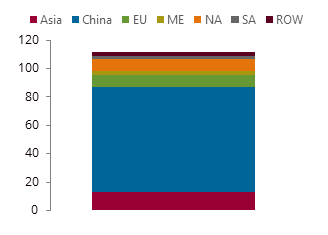
Methanol demand by major region in 2024, kt
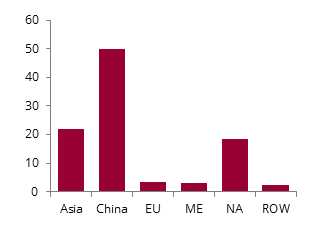
Methanol supply by major region in 2024, %
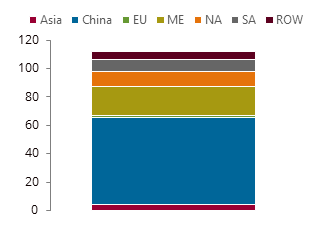
Methanol supply by major region in 2024, kt
Manufacturing of methanol
Methanol is made from synthesis gas (syngas), which is the product of hydrocarbon-rich material that has been heated in the presence of metal catalysts. Almost exclusively, that hydrocarbon is natural gas (methane). However, in China, a large amount of production capacity uses coal as the source of hydrocarbons, so a discussion of natural gas-based and coal-based methanol production processes is merited.
Natural gas-based methanol production
Methanol production from syngas takes place in three major steps: first, reforming the catalysed production of syngas from saturated, de-sulphurised natural gas (reformation); second, methanol synthesis with a Cu/Zn/Alumina catalyst and, finally, crude methanol purification via distillation.
Coal-based methanol production
Coal is the only other significant commercial feedstock for methanol production and its use is concentrated in coal-rich China. The production of syngas from coal has been a commercial reality since the 1920s. The process also follows the three main steps of the natural gas process.
Significant detail regarding methanol production processes, including reformer technologies and comparative natural gas versus coal economics, can be provided separately.





