Fuel cells (using methanol)
Serious development of hydrogen (H2) fuel cells began roughly 60 years ago, with the first important use for the provision of power and water on manned US space flights. There are various benefits of fuel cells, which create power (DC voltage) through an electrochemical reaction that requires H2 and oxygen, yielding water, CO2 and electricity. Over time, methanol has increasingly become a hydrogen source of choice for fuel cells, largely owing to its relatively safe and easy transportation. Methanol can also be made from renewable (aka “low carbon”) sources, so prospects for extremely low-carbon power sources are increasingly in focus.
The renewable methanol power cycle (simplified)
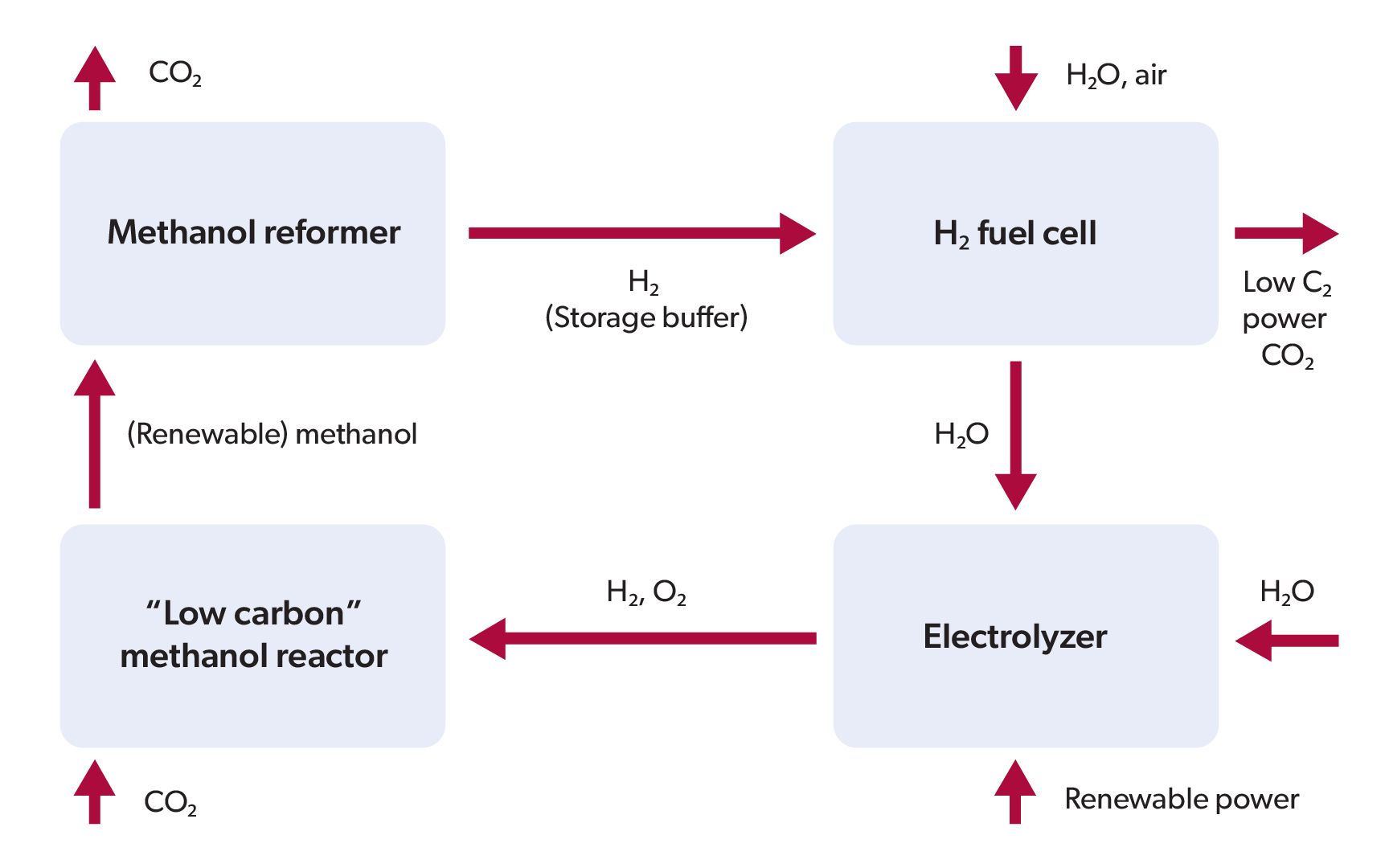
Renewable methanol can be made, reformed to H2 and sent to an H2 fuel cell making low-carbon power using renewable power solutions available today (wind, solar, hydropower, geothermal, tidal etc.) with an electrolyser and water, air and a small amount of net CO2 uptake—CO2 needed to make methanol in the low-carbon methanol reactor should largely offset CO2 produced by the methanol reformer and H2 fuel cell. This power is being developed for stationary or vehicular power generation. Fuel cell and electrolyser production is at a very advanced stage and, by some estimates, over 15,000 fuel cell vehicles have been built. However, many of these installations need a practical source of H2 to be viable, which is the role methanol plays.
Global advances in technology are continually improving the feasibility of fuel cell use in commercial applications.. Companies like Element 1 are licensing advanced methanol reformer process technology, Palcan is making methanol reformer/fuel cell systems, and Blue World Technologies’ reformer and fuel cell process are being incorporated in The Nathalie (pictured below), a Roland Gumpert creation currently under development.






